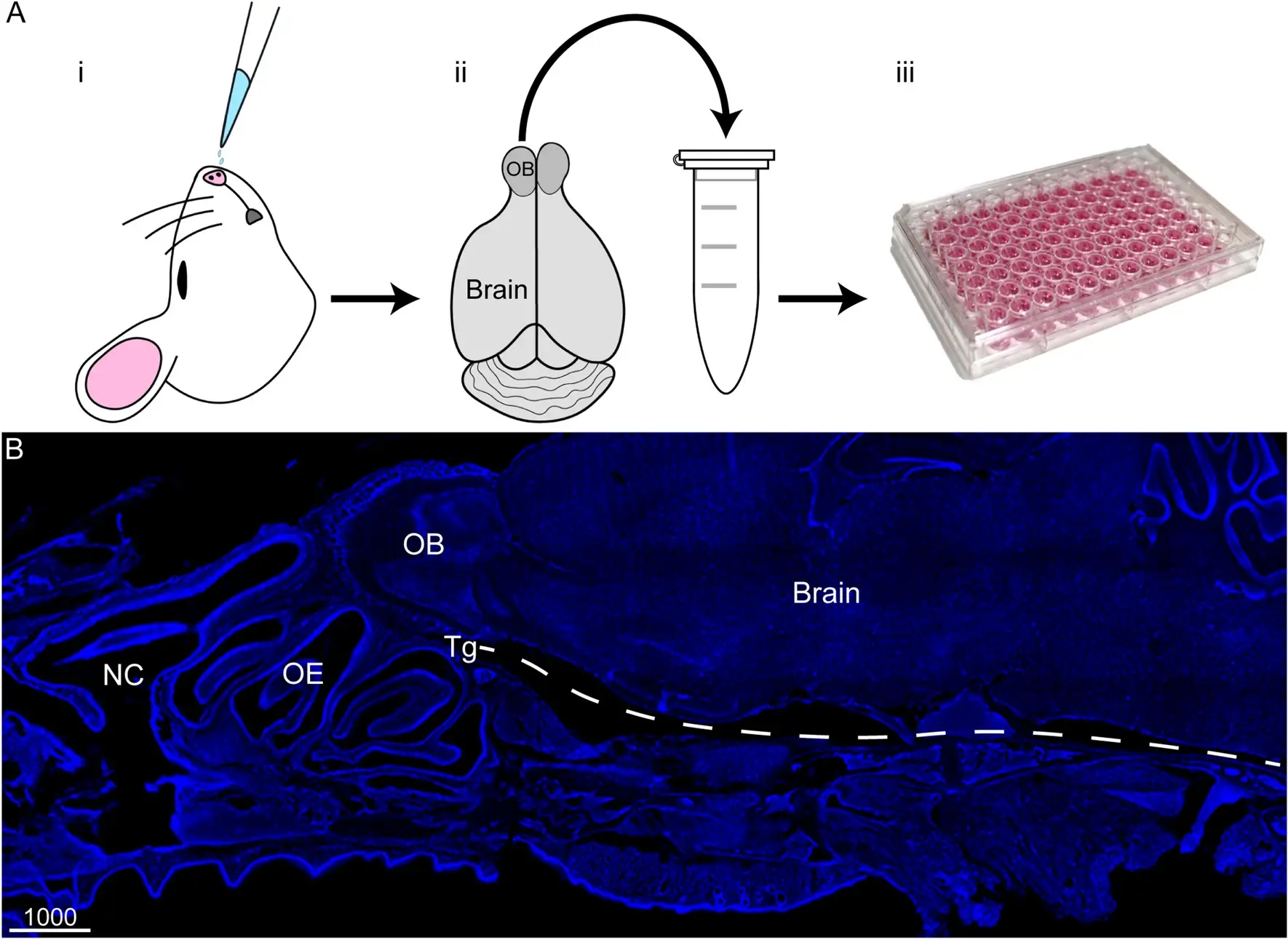
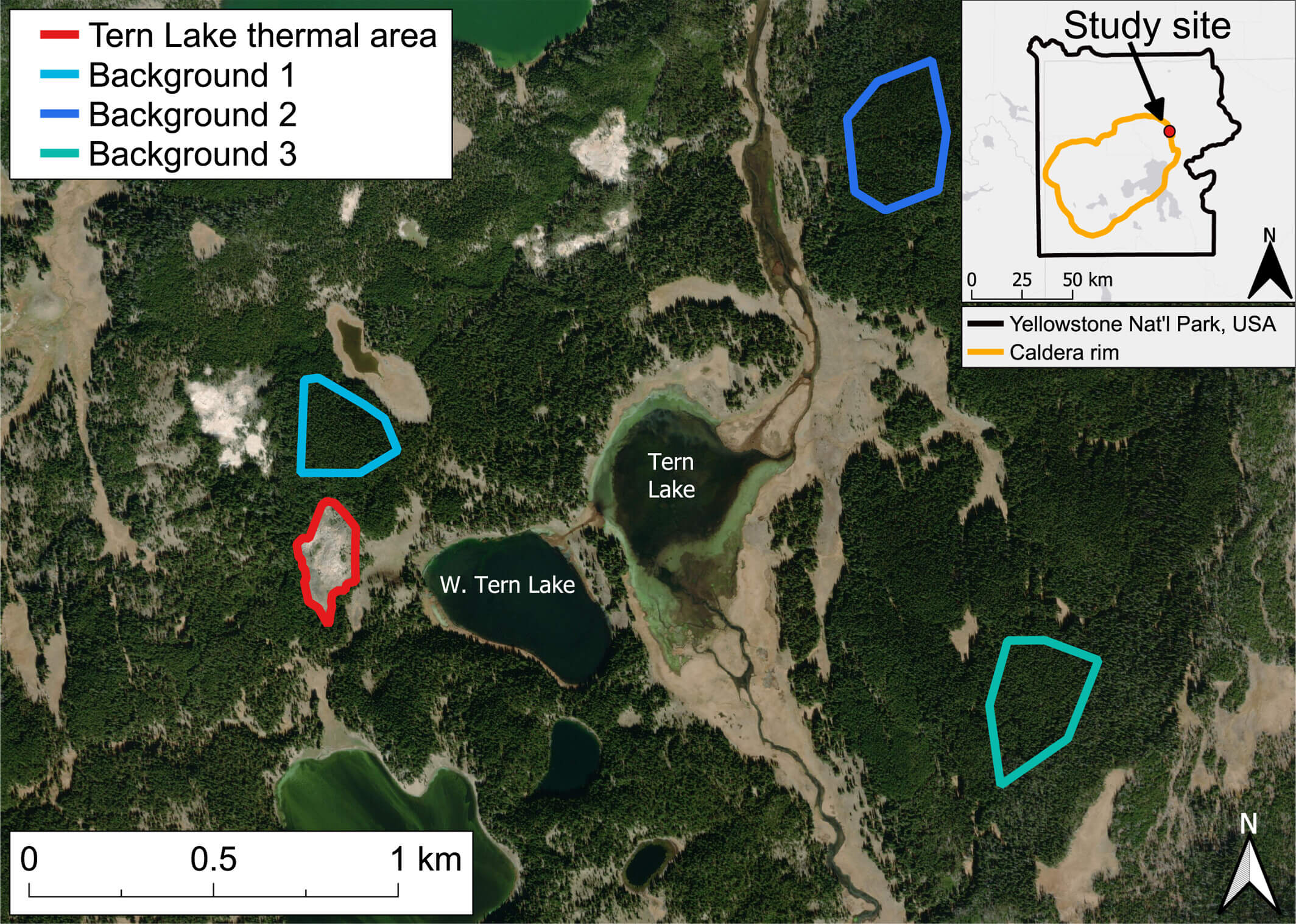
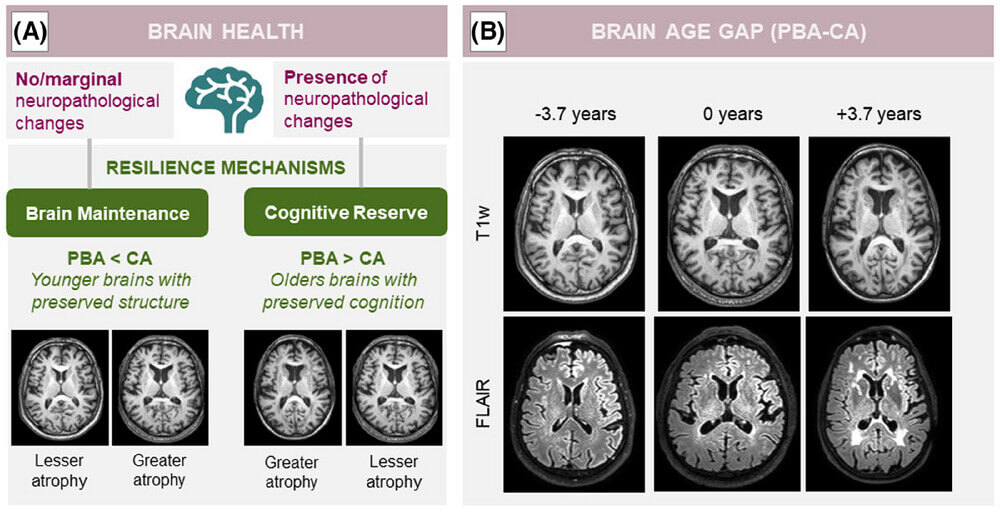
- April 5, 2025
- April 5, 2025
- April 4, 2025
- April 4, 2025
- March 27, 2025
- March 26, 2025
- February 15, 2025
- February 14, 2025
- February 12, 2025
- January 24, 2025
Stoichiometry Quiz – Practice MCQs with Answers
Published On: December 31, 2024Categories: Quizzes
More Research News
- February 10, 2025Lifestyle
©2025 • moleculis.com