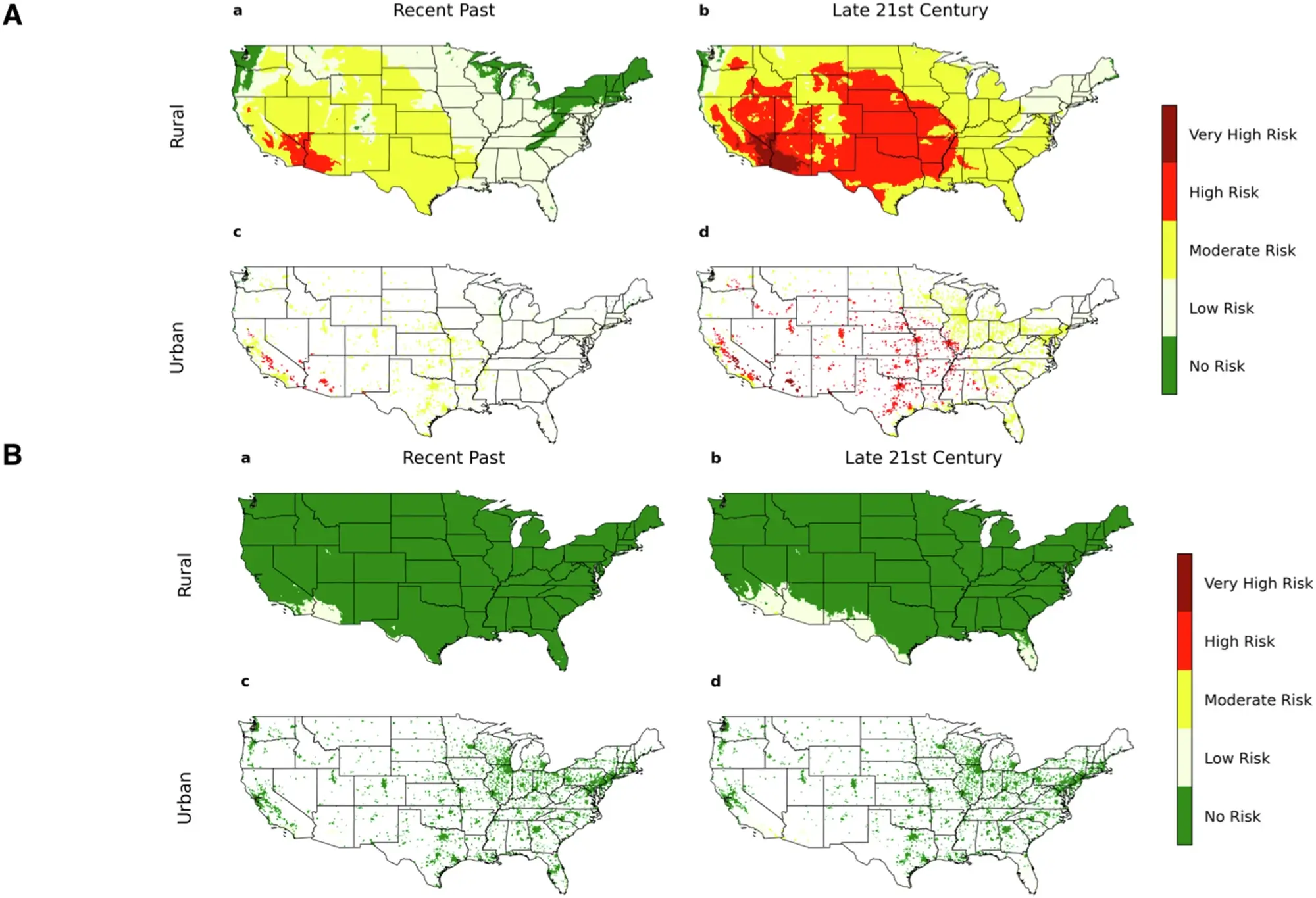
Plastic pollution is no longer just a visible problem washing up on shores—it’s also an invisible threat lurking beneath the surface. Microplastics, tiny fragments of plastic debris less than five millimeters in size, are now found in oceans, lakes, and even drinking water.
Despite efforts to filter them out, these particles are notoriously difficult to remove from large bodies of water. But now, scientists at North Carolina State University may have found an ingenious solution: self-propelling microcleaners that actively seek out and capture microplastics, offering a potential breakthrough in environmental cleanup.
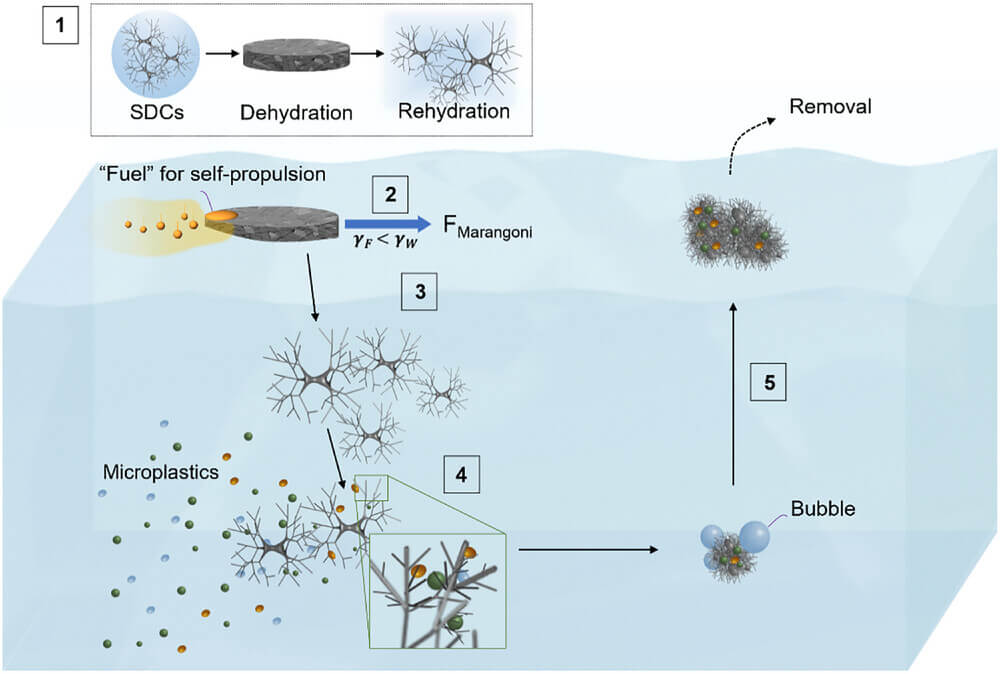
Led by chemical engineer Orlin D. Velev and his team, the research introduces tiny, biodegradable microcleaners capable of navigating water surfaces on their own, gathering microplastics in their path, and then rising to the surface for easy collection. Unlike conventional filtration or centrifugation methods, which struggle to handle the sheer scale of microplastic pollution, these innovative microcleaners operate autonomously, mimicking nature’s efficiency.
The secret lies in their unique design. Made from chitosan, a biodegradable polymer derived from shellfish waste, the microcleaners are structured like tiny branched networks, similar to the microscopic hairs on a gecko’s feet. This allows them to latch onto plastic particles using van der Waals forces—weak but effective intermolecular attractions. In tests, the microcleaners demonstrated an impressive ability to collect a significant portion of dispersed microplastics, even in salty water, where other methods often fail.
One of the most remarkable features of these microcleaners is their mobility. Instead of relying on external energy sources, they propel themselves across the water surface using the Marangoni effect—a phenomenon in which differences in surface tension create motion. Scientists achieved this by embedding small amounts of natural oils like oleic acid or eugenol into the microcleaners. As the oil spreads out, it reduces surface tension and generates movement, allowing the tiny particles to traverse large areas of water, dispersing efficiently and maximizing microplastic capture.
But motion alone isn’t enough; retrieval is just as crucial. The team solved this problem by incorporating magnesium microparticles within the microcleaners. When submerged, these particles react with water, producing hydrogen bubbles that lift the microcleaners back to the surface, carrying their microplastic cargo with them. This self-sustaining cycle of propulsion, capture, and resurfacing sets this method apart from other cleanup strategies.
Laboratory experiments confirmed the microcleaners’ effectiveness. In controlled water samples spiked with fluorescent polystyrene beads—commonly used to simulate microplastic pollution—the microcleaners captured up to 85% of the contaminants. Even in highly saline water, a challenge for many existing filtration technologies, the microcleaners retained their efficiency. “We wanted a system that doesn’t just collect microplastics but also retrieves them in a way that’s scalable and sustainable,” said Velev, emphasizing the practical potential of the technology.
Traditional approaches to microplastic cleanup, such as filtration and skimming, often fail to address smaller particles or require vast energy inputs. Some experimental solutions involve self-propelling micromotors, but these often depend on harmful chemicals like hydrogen peroxide or hydrazine as fuel, making them unsuitable for open-water applications. In contrast, Velev’s microcleaners use environmentally friendly materials, ensuring they don’t introduce new pollutants into the ecosystem.
Reference
https://doi.org/10.1002/adfm.202423494