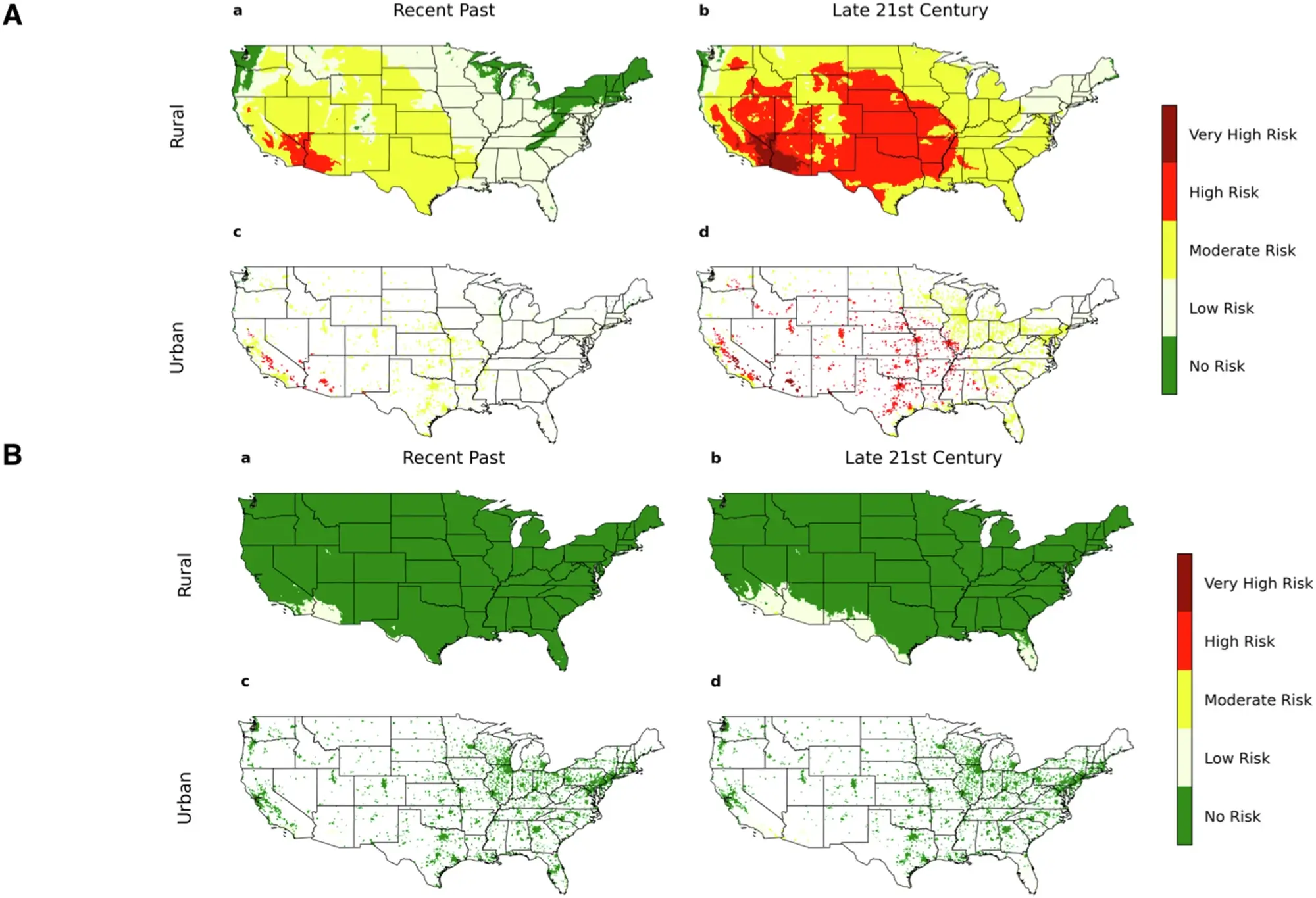
Easily determine the percent composition of an element in a compound using our Percent Composition Calculator. This tool supports unit conversions for grams (g), milligrams (mg), kilograms (kg), pounds (lb), and ounces (oz), making your calculations accurate and effortless.
What Is Percent Composition?
Percent composition is the percentage of an element’s mass relative to the total mass of the compound. It helps chemists and researchers determine the relative amounts of elements in a substance.
The formula for percent composition is:
Percent Composition = (Mass of Element / Mass of Compound) × 100
This formula ensures that the contribution of each element to a compound is properly quantified.
How to Calculate Percent Composition
To calculate percent composition, follow these steps:
- Enter the mass of the element in your desired unit (g, mg, kg, lb, or oz).
- Enter the mass of the compound in the same or a different unit.
- The calculator automatically converts all units to grams (g) before performing the calculation.
- The percent composition is calculated and displayed with the appropriate unit.
Step-by-Step Percent Composition Calculation
Let’s calculate the percent composition of oxygen in water (H₂O).
Step 1: Write the Formula
Percent Composition = (Mass of Element / Mass of Compound) × 100
Step 2: Find the Molar Mass of Water (H₂O)
To determine the mass of each element in water, we need the atomic masses from the periodic table:
- Hydrogen (H) = 1.008 g/mol
- Oxygen (O) = 16.00 g/mol
Water (H₂O) consists of two hydrogen atoms and one oxygen atom, so its molar mass is:
Molar Mass of H₂O = (2 × 1.008) + (1 × 16.00)
= 2.016 + 16.00
= 18.016 g/mol
Step 3: Calculate the Mass Contribution of Oxygen
Mass of Oxygen in H₂O = 16.00 g/mol
Step 4: Apply the Formula
Percent Composition of Oxygen = (Mass of Oxygen / Mass of H₂O) × 100
= (16.00 / 18.016) × 100
= 88.81%
Final Answer
Oxygen makes up 88.81% of the mass of water (H₂O).
Example Calculations
Example 1: Using the Same Unit
Given:
- Mass of element = 20 grams (g)
- Mass of compound = 80 grams (g)
Calculation:
(20 g / 80 g) × 100 = 25.0000%
Example 2: Using Different Units
Given:
- Mass of element = 50 pounds (lb)
- Mass of compound = 2 kilograms (kg)
Unit Conversion:
- 50 lb = 22,679.6 g
- 2 kg = 2,000 g
Calculation:
(22,679.6 g / 2,000 g) × 100 = 1,133.9800%
Example 3: Small Quantities
Given:
- Mass of element = 500 milligrams (mg)
- Mass of compound = 5 grams (g)
Unit Conversion:
- 500 mg = 0.5 g
Calculation:
(0.5 g / 5 g) × 100 = 10.0000%
Why Use This Calculator?
Supports multiple unit conversions (grams, mg, kg, lb, oz)
Instant results with correct unit formatting
Great for chemistry, pharmacy, and material science calculations
Accurate and optimized for real-world applications
Use our Percent Composition Calculator now to simplify your calculations and save time!