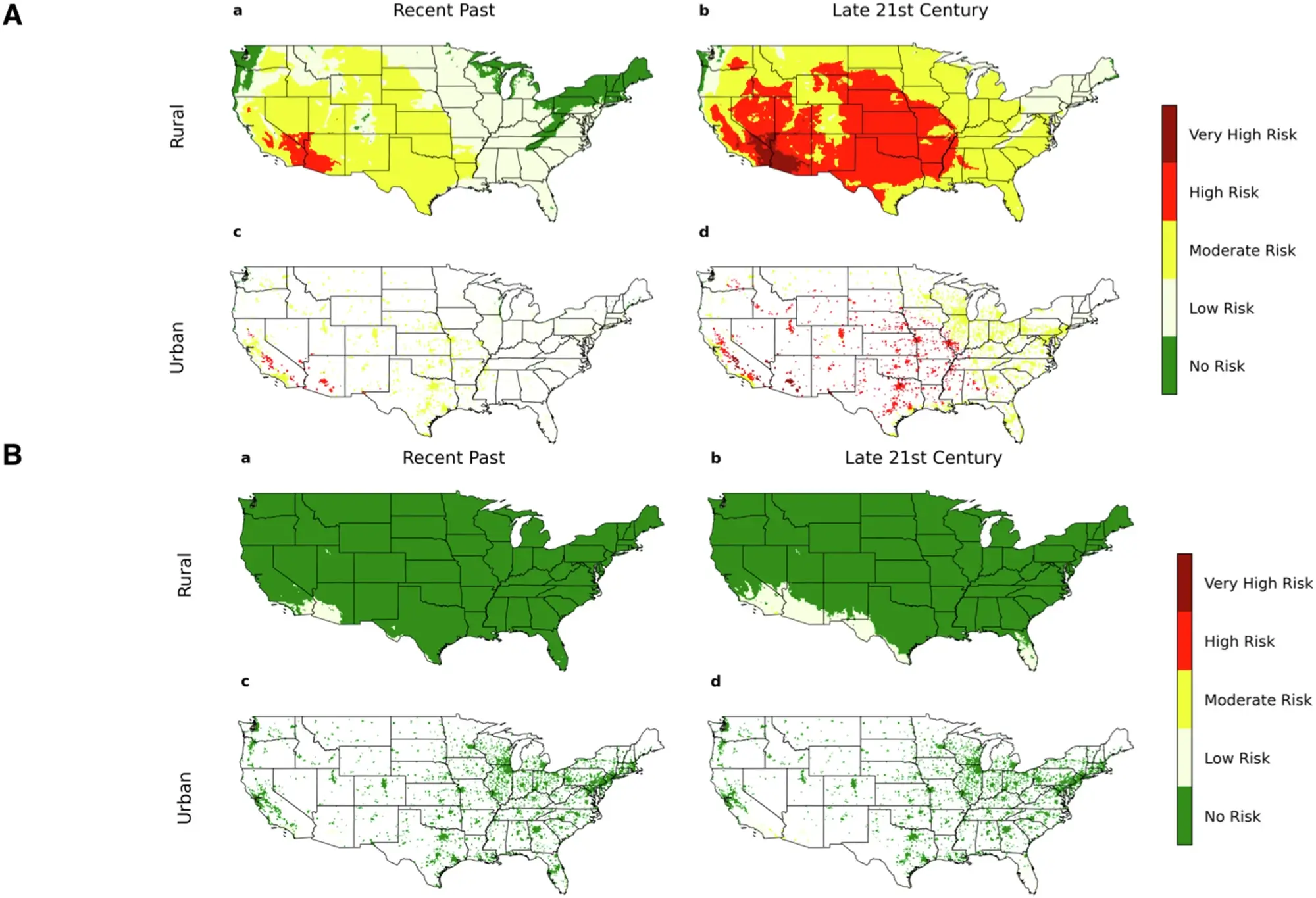
What is Normality?
Normality (N) is a measure of concentration in chemistry that indicates the number of equivalents of a solute present in one liter of solution. It is commonly used in acid-base reactions, redox reactions, and titrations.
Formula for Normality
The most common formula to calculate normality is:
Normality (N) = Mass of Solute (g) / (Equivalent Weight (g/eq) × Volume of Solution (L))
Steps to Calculate Normality
- Determine the mass of the solute (g).
- Find the equivalent weight (g/eq) of the solute using its molecular weight and valency.
- Measure the volume of the solution in liters (L).
- Apply the formula and calculate the normality.
Different Ways to Calculate Normality
1. Using Mass, Equivalent Weight, and Volume
This is the standard method using the formula:
N = Mass (g) / (Equivalent Weight (g/eq) × Volume (L))
Example Calculation:
- Given: Mass = 2 g, Equivalent Weight = 40 g/eq, Volume = 500 mL (0.5 L)
- Calculation: N = 2 / (40 × 0.5) = 2 / 20 = 0.1 N
2. Using Molarity and Valency
If molarity (M) of the solution is known, normality can be calculated using:
N = Molarity (M) × Valency
Example Calculation:
- Given: Molarity = 0.5 M, Valency = 2
- Calculation: N = 0.5 × 2 = 1.0 N
3. Using Number of Equivalents and Volume
If the number of equivalents of solute is given, normality is calculated as:
N = Number of Equivalents / Volume (L)
Example Calculation:
- Given: Number of Equivalents = 0.02 eq, Volume = 250 mL (0.25 L)
- Calculation: N = 0.02 / 0.25 = 0.08 N
Key Takeaways
- Normality measures the reactive concentration of a solution.
- It depends on the equivalent weight, which varies based on the reaction type.
- Multiple methods exist to calculate normality, depending on available data.
- Unit conversions (mg to g, mL to L) are crucial for accurate results.