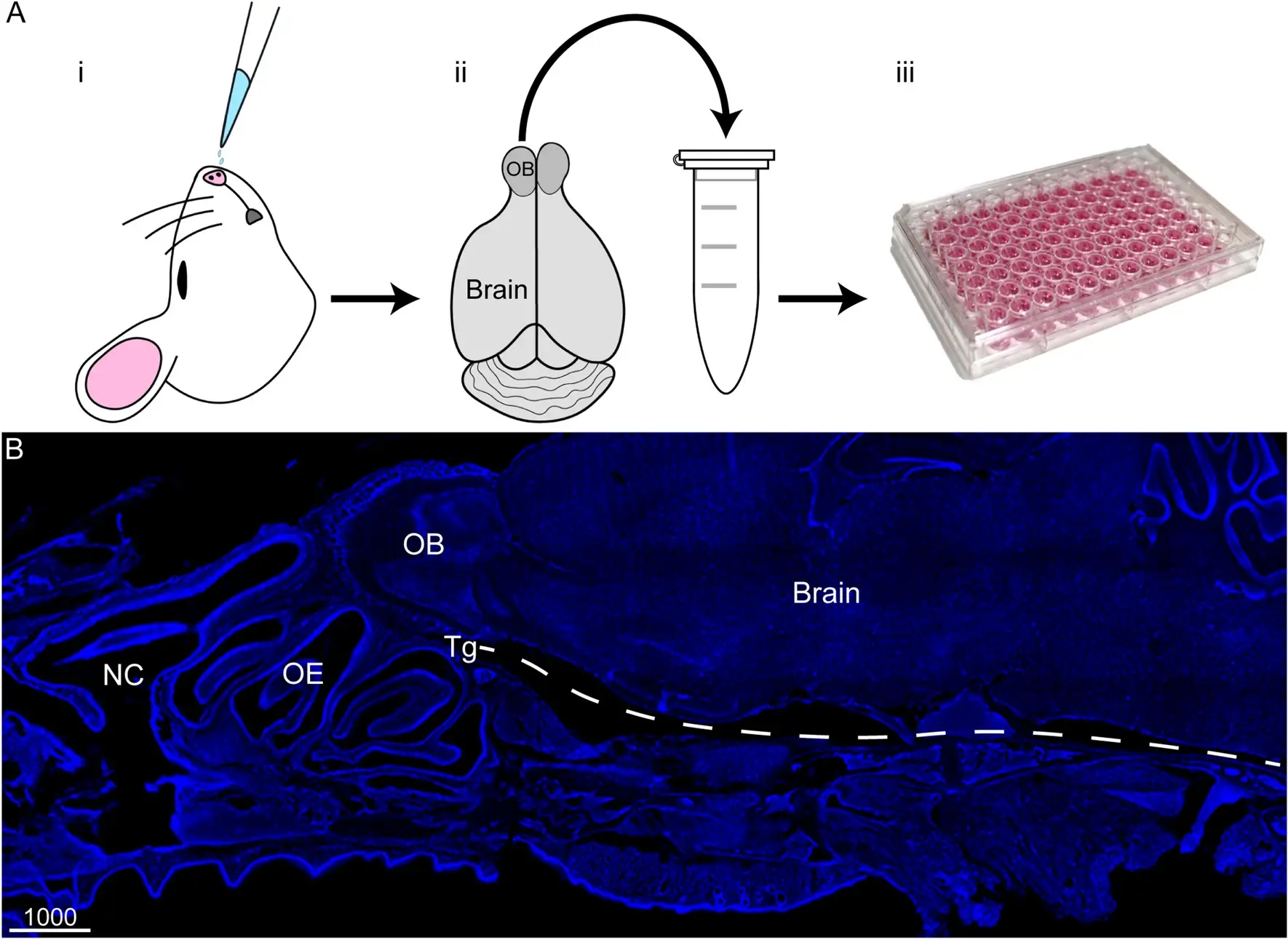
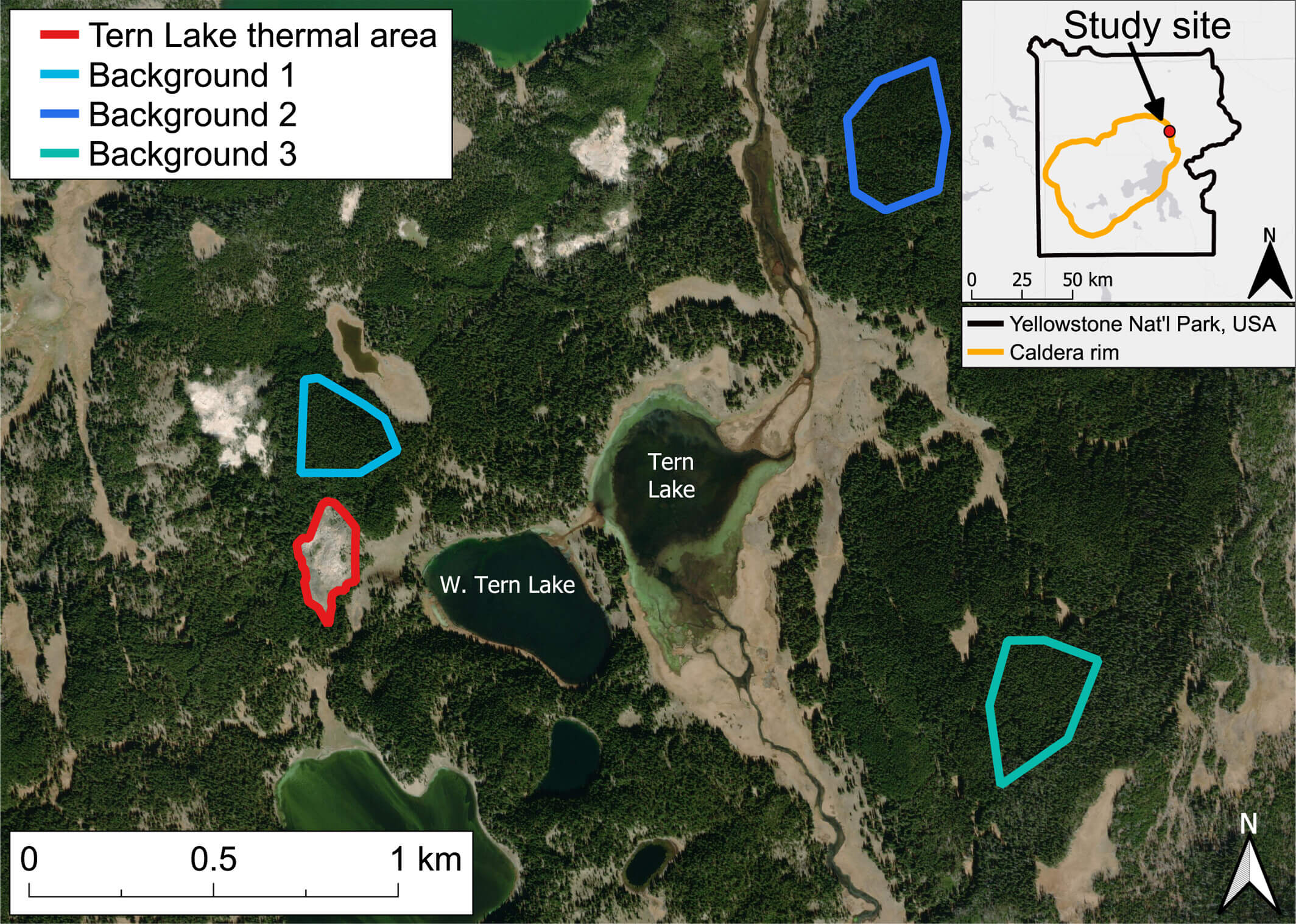
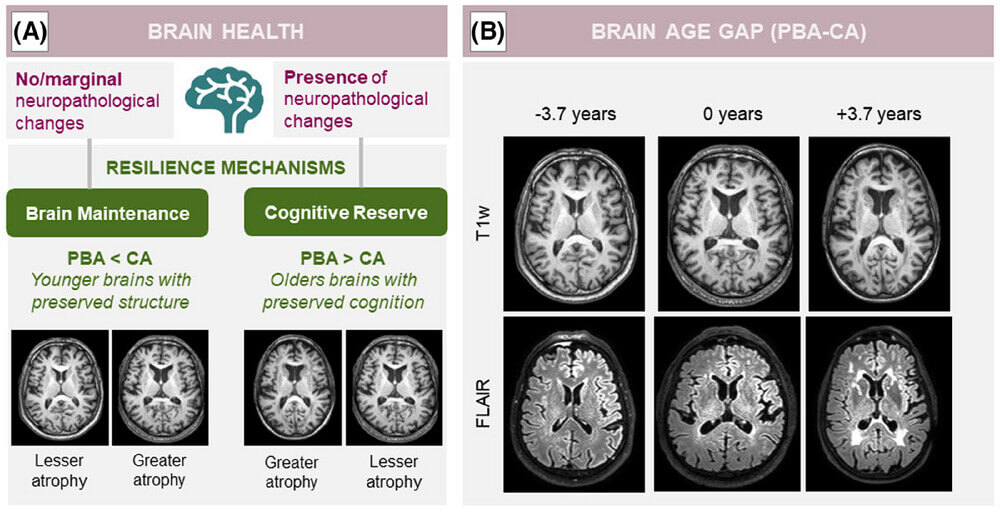
What to Expect in the Quiz
- Conceptual Questions: Assess your understanding of bonding, anti-bonding, and non-bonding orbitals.
- Calculations: Determine bond order and magnetic properties of various diatomic molecules.
- Electronic Configurations: Identify correct molecular orbital diagrams and electron arrangements.
- Comparative Analysis: Analyze stability, bond length, and energy levels of molecular species.
Each question is followed by a detailed explanation, ensuring you not only test your knowledge but also learn from any mistakes.